Pancreatic Ductal Adenocarcinoma (PDAC) remains a formidable clinical challenge, characterized by a highly immunosuppressive tumor microenvironment (TME) and resistance to conventional therapies. While Irreversible Electroporation (IRE) offers a non-thermal ablation option that preserves critical tissue structures, its efficacy is limited by the immunologically "cold" nature of PDAC. This cold TME—defined by scarce immune infiltration and defective antigen presentation—facilitates immune evasion and rapid recurrence. Although combining IRE with immunoadjuvants like CpG ODN (a TLR9 agonist) is a promising strategy, the clinical translation of CpG ODN is hindered by enzymatic degradation, poor biodistribution, and systemic toxicity. Furthermore, existing nanomedicine platforms often struggle with premature drug release or lack responsiveness suitable for deep-seated tumors. Consequently, designing an intelligent, tumor-targeted, and IRE-responsive nano-platform is critical to achieving synergistic ablation and immunotherapy.
In a study published in Advanced Materials, Prof. Tian’an Jiang from the First Affiliated Hospital, Zhejiang University School of Medicine, presented a transformative solution to these challenges. The research team developed a novel nano-immunoadjuvant system (PSFC) that integrates pancreatic cancer targeting peptides, superparamagnetic iron oxide, ferrocene (Fc), and CpG ODN. This "electro-immunotherapy" platform is designed to be triggered specifically by the IRE electric field, enabling precise spatiotemporal control over immunoadjuvant release.
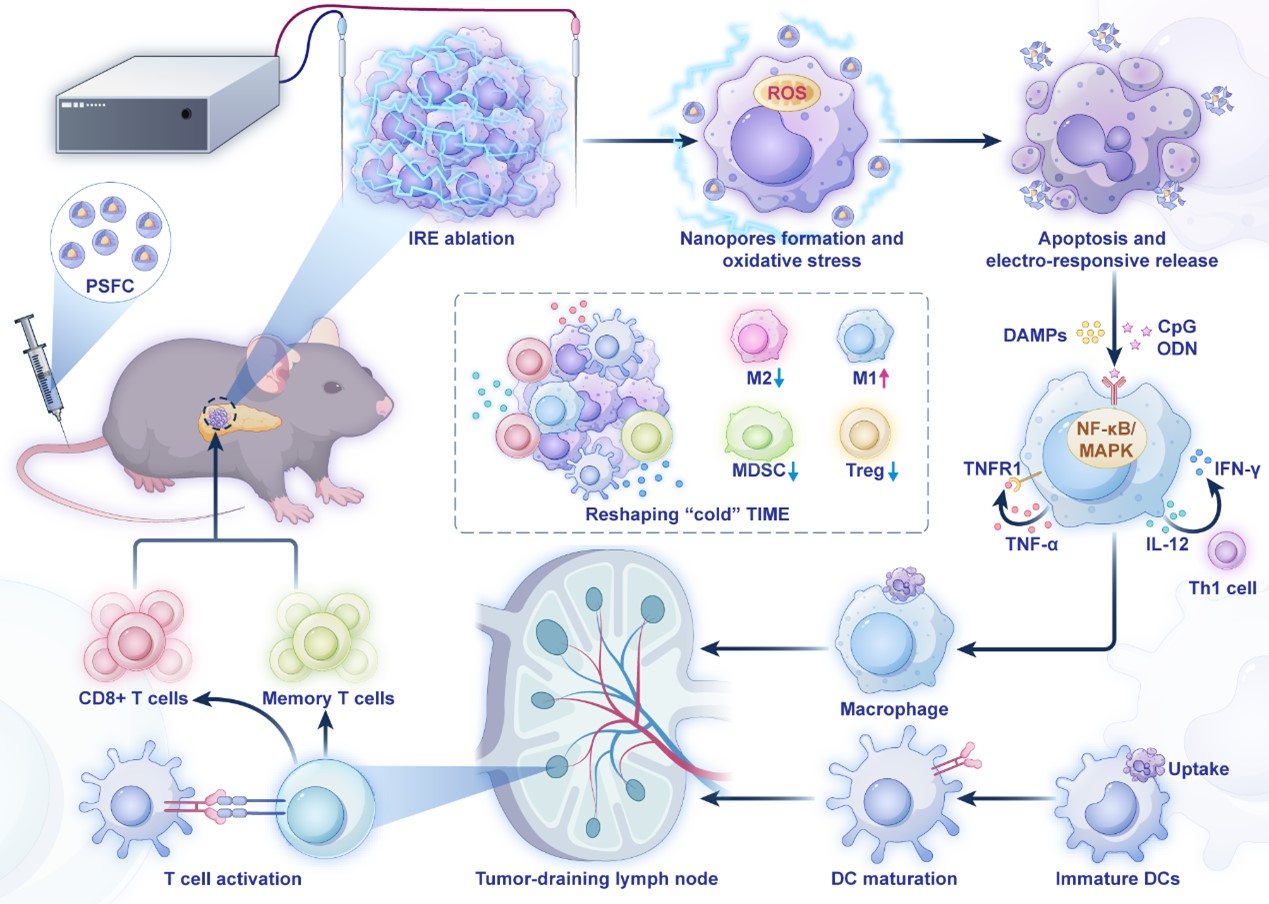
Fig.1 Schematic illustration of macrophage-associated Th1-immune response induced by electro-immunotherapy in the treatment of Pancreatic ductal adenocarcinoma.
The study first elucidated the biphasic kinetic profile of the post-IRE immune microenvironment. Using high-dimensional flow cytometry and cytokine analysis, researchers discovered that while IRE induces transient early immune activation, the TME rapidly reverts to an immunosuppressive state due to PDAC’s intrinsic stromal barriers. This "early activation, late suppression" dynamic underscores the necessity of combination therapy. The PSFC system was engineered to address this window of opportunity. Physicochemical characterization confirmed its excellent electrochemical dissociation properties and MRI contrast capabilities, allowing for non-invasive monitoring. In vivo imaging demonstrated that PSFC achieves deep tumor penetration and high accumulation via peptide-mediated targeting.
Mechanistically, the combination of IRE and PSFC synergistically induces Immunogenic Cell Death (ICD). In vitro assays confirmed that the therapy significantly enhances tumor cell killing by generating Reactive Oxygen Species (ROS) and releasing Damage-associated Molecular Patterns (DAMPs). Crucially, the IRE-triggered release of CpG ODN stimulates the maturation of antigen-presenting cells, initiating a specific anti-tumor immune response. In murine models of in situ pancreatic cancer, this strategy remodeled the immunosuppressive "cold" TME: it reduced immunosuppressive cells (M2-TAMs, MDSCs, Tregs) while significantly increasing tumor-infiltrating T cells. This resulted in inhibited tumor growth, prolonged survival, and the establishment of potent long-term immune memory that prevented recurrence in re-challenge models.
At the molecular level, the study identified the TLR9-MyD88-NF-κB/MAPK pathway as the driver of this immune reprogramming. The released CpG ODN activates macrophages, inducing the secretion of pro-inflammatory cytokines (IL-12, TNF-α). IL-12 promotes Th1 differentiation and IFN-γ secretion, which in turn drives M1 polarization and CD8+ T cell expansion, creating a positive feedback loop that converts "cold" tumors into "hot" tumors.
In summary, this research establishes PSFC as a pioneering electro-immunotherapy platform that bridges physical ablation and immunomodulation. By overcoming the limitations of CpG ODN delivery and breaking immune tolerance, this strategy offers a clinically translatable blueprint for treating refractory solid tumors. The integration of MRI guidance, deep tissue responsiveness, and potent immune activation positions electric-responsive nanomedicine as a powerful new frontier in oncology, offering hope for improving long-term survival in PDAC patients.
More information: Ph.D. candidate Chengyue Zhang and Dr. Baohua Wang are the co-first authors of this article. Prof. Tian’an Jiang, Prof. Qiyu Zhao, Prof. Xiongbin Lu and Dr. Liting Xie are the co-corresponding authors of this article.


